- Blog
- Edison 5
- Sharp digital alarm clock with jumbo 2 display
- How to disable excel add ins
- Vray maya 2017
- Lola bunny pop vinyl
- Best alarm clock app for android
- Rhonda long hair salon texas
- 99 names of muhammad s-a-w
- How to calculate ppm with molarity
- Pureedge viewer issue saving
- City building games like anno
- Huniepop 2 gameplay
- Classic minecraft resource pack
- Malena movie hindi dubbed
This tool will calculate the molarity of a solution for a compound of known molecular weight. rather than, as these calculators use decimal points for input/output of calculations. Thus for the 1 M NaCl, the total ion concentration is 2 M for the 1 M CaCl 2, the total ion concentration is 3 M. Note: When entering decimal values in the Biomath calculators, please use a decimal point.

In addition, the total ion concentration is the sum of the individual ion concentrations. However, if the solution were 1 M CaCl 2, there are two Cl −(aq) ions for every formula unit dissolved, so the concentration of Cl −(aq) would be 2 M, not 1 M. For example, if 1 M NaCl were prepared, the solution could also be described as a solution of 1 M Na +(aq) and 1 M Cl −(aq) because there is one Na + ion and one Cl − ion per formula unit of the salt. Because the ions in ionic compounds go their own way when a compound is dissolved in a solution, the resulting concentration of the ion may be different from the concentration of the complete salt. What mass of Fe 3+ ion is present in 3,450 mL of H 2O, which has a density of 1.00 g/mL?įor ionic solutions, we need to differentiate between the concentration of the salt versus the concentration of each individual ion. This result compares well to our ballpark estimate (it’s a bit less than one-half the stock concentration, 5 M).The concentration of Fe 3+ ion in a sample of H 2O is 335.0 ppm. The relative amount of sugar in a beverage determines its sweetness (Figure \(\PageIndex=2.36\:M \nonumber\]

The relative amount of the active ingredient in a medicine determines its effectiveness in achieving the desired pharmacological effect. The relative amounts of iron, carbon, nickel, and other elements in steel (a mixture known as an “alloy”) determine its physical strength and resistance to corrosion. The relative amount of oxygen in a planet’s atmosphere determines its ability to sustain aerobic life. Similar to a pure substance, the relative composition of a mixture plays an important role in determining its properties. However, mixtures-samples of matter containing two or more substances physically combined-are more commonly encountered in nature than are pure substances. In preceding sections, we focused on the composition of substances: samples of matter that contain only one type of element or compound.
#HOW TO CALCULATE PPM WITH MOLARITY HOW TO#
Here is how to calculate the concentration. Go here for ISE molarity/ppm conversions shown in Table III. It is the number of moles of target substance (solute) dissolved in 1 liter of solution.
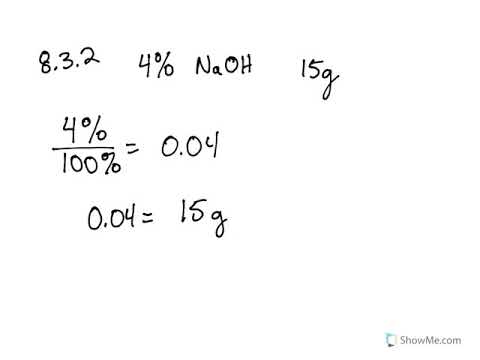
Fluoride has a FW of 19, hence a 10-3 M concentration is equal to 19ppm, 1M is equal to 19,000 ppm and 1ppm is equal to 5.2 x 10-5 M. Calculate the molarity (mol/L) if there is 70 ppm of Ca2+ ions. The FW of an ion species is equal to its concentration in ppm at 10-3 M. Molarity ((ppm 0.001) / molar mass) Example.
